The most dangerous misconception about closed-loop insulin systems is that they “take care of the diabetes.” They absolutely do not. They automate the math, not the medicine.
If you are a clinician and you do not understand the basic logic of these algorithms, you are flying blind when something goes wrong at 2 a.m., when your patient blames the pump, or when a family quietly turns everything to manual because they no longer trust the system.
Let me break this down specifically.
1. What “Closed-Loop” Actually Means (And What It Does Not)
Every glossy brochure and rep talk will tell you “artificial pancreas,” “full automation,” “hands off.” That is marketing. The reality is more constrained and algorithm-dependent.
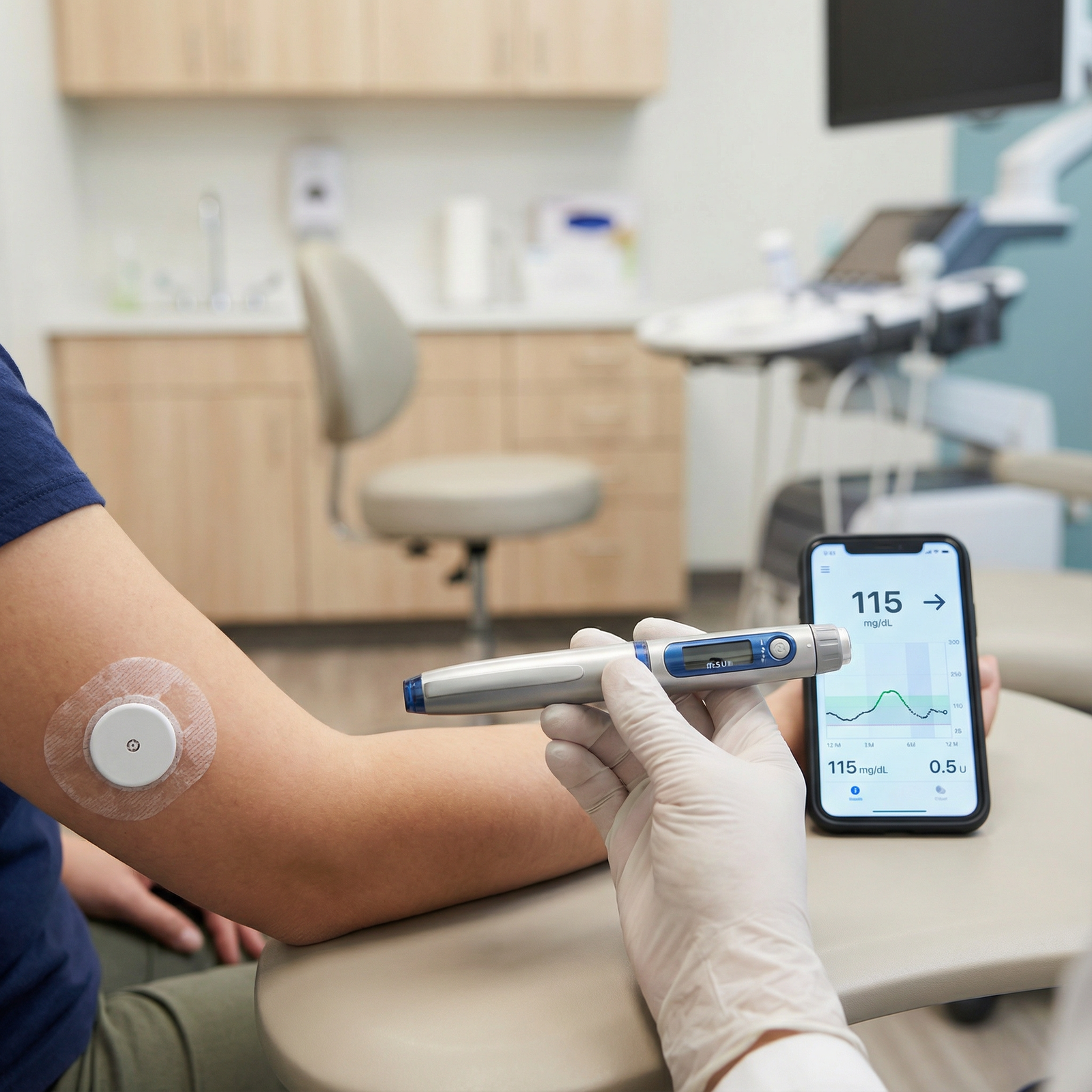
Closed-loop insulin systems (hybrid closed-loop in practice) are essentially three components:
- Continuous glucose monitor (CGM)
- Insulin pump (usually subcutaneous, rapid-acting analogue)
- Control algorithm (on the pump, a phone, or a dedicated controller)
The loop is “closed” because:
- The CGM continuously measures glucose (interstitial, not blood).
- Those readings feed into an algorithm.
- The algorithm automatically adjusts insulin delivery (usually basal or micro-boluses) without the user pressing a button every time.
Where the marketing slides get quietly vague:
- Most commercial systems are “hybrid” closed loop.
The user still has to announce meals / give boluses. - Insulin is still subcutaneous with slow kinetics. No algorithm can fully compensate for late boluses, skipped boluses, or erratic eating.
- Algorithms are conservative by design—safety over tight control.
So no, this does not replace diabetes self-management. It changes the work:
- Less micromanaging basal rates
- More focus on carb counting timing
- More focus on understanding system limits and failure modes
And that is where your understanding of the logic becomes critical.
2. The Core Algorithm Archetypes: How the System “Thinks”
Different companies will give you different brand names, but under the hood they are all variations on a few core control strategies. If you can recognize these patterns, you can predict system behavior.
2.1 Three Big Families of Control Logic
| Algorithm Type | Commercial Examples | Key Concept |
|---|---|---|
| PID Control | Early research systems, some legacy designs | Reacts to current, past, and rate-of-change of glucose |
| Model Predictive Control (MPC) | Medtronic 670G/770G (hybrid), in some research systems | Predicts future glucose and optimizes insulin over a horizon |
| Fuzzy / Heuristic / Adaptive | DIY Loop, AndroidAPS, Control-IQ elements | Rule-based with adaptive sensitivity and constraints |
The average clinician does not need to derive control equations, but you should know how each “thinks” operationally.
PID (Proportional–Integral–Derivative) control
Conceptually:
- Proportional: Responds to how far the glucose is from target.
- Integral: Responds to how long it has been off target (cumulative error).
- Derivative: Responds to how fast glucose is changing.
Clinical translation:
- Big jump above target → more insulin
- Staying above target for hours → gradually ramps insulin more
- Glucose rising quickly → more aggressive; dropping quickly → backs off
This is intuitive but can be noisy if not well tuned. Many modern products have moved to more sophisticated approaches.
Model Predictive Control (MPC)
This is the “chess player” of algorithms.
- Uses a mathematical model of glucose–insulin dynamics.
- Predicts glucose 30–120 minutes into the future.
- Chooses an insulin profile over that horizon that will keep predicted glucose near a target, under dosing constraints.
- Repeats this every few minutes with updated data.
Clinically, MPC systems may do things that look weird if you do not understand them:
- They may keep basal high even when current glucose is okay, because the model predicts a rise.
- Or they may cut insulin early, anticipating upcoming lows from past boluses or exercise.
Fuzzy / heuristic / adaptive logic
This is closer to how an experienced patient behaves.
- Rule-based: “If CGM rising fast after meal, increase micro-boluses.”
- Uses patterns: updates insulin sensitivity or carb ratio based on past data.
- Puts strict guards against hypoglycemia and insulin stacking.
DIY systems like Loop / AndroidAPS, and some commercial ones (Tandem Control-IQ) use hybrid approaches:
- A predictive component (like a simple MPC)
- Plus safety constraints
- Plus adaptation of patient-specific parameters over time
You will often see this described as “adaptive” or “learning” systems.
3. The Single Most Important Concept: The Target and Its Safety Envelope
Everything in closed-loop logic orbits one thing: the target glucose and how aggressively you are allowed to move toward it.
Most systems do not shoot for 90 mg/dL. That would be unsafe with current insulin kinetics. Targets are usually in the 100–120 mg/dL range, with explicit lower safety bounds.

| Category | Value |
|---|---|
| Nominal Target | 110 |
| Minimum Allowed | 80 |
| Suspend Threshold | 70 |
Key points you must know:
- Nominal target: The value the algorithm aims for over hours.
- Safety floor: A glucose value below which aggressive insulin cuts begin.
- Suspend threshold: Value at which insulin delivery may be suspended entirely (often ~70 mg/dL or based on predicted lows).
What this means clinically:
- Pushing the target too low (in systems that allow it) will usually increase alarms, autobasal cuts, and “bouncing” from lows to reactive highs.
- Systems with fixed targets (e.g., older Medtronic 670G ~120 mg/dL) will feel “too high” for some patients but are designed that way to reduce severe hypos.
- “Sleep” or “exercise” targets are not gimmicks; they change the safe operating envelope.
And here is the ethical piece: if you do not understand how the target and safety limits interact, you will not give informed recommendations. You will either promise too much (“this will get you to 90–110 always”) or too little (“it is not really any better than a pump + CGM”).
4. How the Algorithm Uses Inputs You Control (And Miscontrol)
Closed-loop does not operate on CGM alone. It heavily relies on:
- Basal rate settings (even if they are “hidden” in some systems)
- Insulin-to-carbohydrate ratio
- Correction factor (insulin sensitivity factor)
- Insulin action time (duration of insulin effect)
- Announced carbs / meal boluses
- Declared modes (sleep, exercise, etc.)
These parameters are not ornamental. They define the algorithm’s belief about the patient. When you change them blindly, you distort how the system “thinks.”
4.1 The quiet giant: insulin action time (IAT or DIA)
This is one I routinely see mis-set by clinicians.
If you set insulin action time too short (e.g., 2 hours for lispro/aspart), the algorithm believes insulin disappears quickly.
→ It is more willing to give additional insulin earlier.
→ Higher risk of late hypoglycemia due to hidden stacking.If you set it too long (e.g., 6–7 hours), the algorithm believes a lot of insulin is still active.
→ It under-corrects highs.
→ Patients blame the algorithm for being “lazy.”
Reality: For rapid-acting analogues, most modern systems assume ~4–6 hours of tail, not 2–3. The visible drop may be quick, but the tail matters a lot for safe automation.
4.2 Insulin sensitivity and carb ratio in an automated world
Gut reaction from many clinicians: “The algorithm adapts, so these matter less.” Wrong.
- In many systems, ISF and ICR are still the starting point for corrections and meal boluses.
- The closed-loop algorithm modulates around those numbers; it does not fully override them.
Clinical pattern you will see:
- If patient is routinely high after meals but nighttime control is excellent on auto mode → ICR often too weak (too few units per carb).
- If patient crashes 2–3 hours after every correction bolus while auto mode is on → ISF too strong (too big of a glucose drop per unit).
The key nuance: do not “fix” these by trying to fight the closed-loop settings. Work with them. Adjust the foundational pump parameters first; then see how the closed loop behaves.
5. Boluses, Basal Modulation, and Why Postprandial Spikes Still Happen
Let me kill a myth: No current commercial closed-loop system can reliably “catch up” to large unannounced or very late-announced meals with standard rapid-acting insulin.
Subcutaneous insulin is too slow.
So the typical control architecture splits responsibilities:
- Basal (or micro-boluses every 5–10 minutes): Algorithm-controlled, gradually adjusting up or down.
- Meal and correction boluses: User-initiated, sometimes algorithm-assisted (e.g., automated corrections).
What the algorithm actually does during a meal:
- User enters carbs and gives a bolus (in hybrid systems).
- Algorithm watches the CGM trajectory.
- If glucose is rising faster than expected, and insulin on board is “insufficient” per its model, it may:
- Add small automatic corrections
- Temporarily increase basal
- If glucose is dropping faster than expected, it may:
- Reduce or suspend basal
- In some research systems, suggest carb intake
So why do patients still spike to 250–300 mg/dL?
- Late boluses (bolus after eating, not before).
- Underestimated carbs.
- Very high-glycemic meals (rapid absorption beats subcutaneous insulin onset).
- Fear of hypoglycemia leading to intentionally small boluses.
Your role is to explain this honestly:
- The algorithm will usually reduce time >250 mg/dL.
- It will not “erase” a huge late meal bolus.
- Pre-bolusing and realistic carb counting still matter.
If you oversell the system, patients lose trust when these very predictable spikes still occur.
6. Safety Logic: How Systems Try to Prevent Hypoglycemia
Here is where the algorithms earn their regulatory approval.
Broadly, they use a combination of:
- Real-time CGM reading
- Rate-of-change (slope) of glucose
- Insulin on board estimates
- Prediction of future glucose (15–60+ minutes ahead)
Common safety mechanisms:
- Low-glucose suspend (LGS): Suspends insulin when glucose is below or approaching a threshold (e.g., 70–80 mg/dL).
- Predictive low-glucose suspend (PLGS): Suspends insulin when predicted glucose in the near future will cross a threshold, even if current value is still normal.
- Automated basal reduction: Gradually dials down basal as predicted lows approach, not just full suspension.
- Hypo-avoidant targets: During sleep or exercise, the algorithm raises targets and thresholds.

| Category | Value |
|---|---|
| Manual Pump+CGM | 60 |
| Predictive Suspend | 70 |
| Full Hybrid Closed Loop | 75 |
Interpretation: as you move from manual pump+CGM to predictive suspend to hybrid closed loop, time in range tends to rise, but only because hypoglycemia is aggressively avoided alongside smarter insulin delivery.
Clinical scenarios you will see:
Patient waking up high (180–220 mg/dL) with “graph looks like a cliff at 3 a.m.”
→ Algorithm saw a predicted low, suspended insulin, then patient rebounded.
→ Usually due to overly aggressive basal or strong correction factor at night.Patient reports “pump turned off my insulin for hours, I do not trust it.”
→ Look at CGM; often there was a legitimate predicted low.
→ Or there are calibration errors / compression lows tricking the system.
This is where your understanding of the prediction logic matters clinically and ethically. It allows you to:
- Reassure appropriately when the system behaved correctly.
- Recognize when sensor issues or parameter mis-settings are driving over-suspension.
- Prevent knee-jerk disabling of safety features that actually reduce severe hypos.
7. Failure Modes, Edge Cases, and Blame
Closed-loop systems do fail. Not dramatically, usually. They fail quietly, through:
- Sensor inaccuracies
- Occlusions or site failure
- Incorrect parameters (e.g., wrong weight, wrong insulin type)
- Overriding or bypassing the algorithm too often
Ethically, how you frame responsibility here matters.
7.1 Sensor issues
CGM input is the foundation. Garbage in, garbage out.
Common patterns:
- Compression lows (patient lies on sensor at night) → Algorithm cuts insulin → Rebound highs.
- Calibration errors → System chronically “believes” wrong values, mis-estimates insulin needs.
Your job:
- Teach patients to recognize improbable CGM readings (e.g., 40 mg/dL but talking, walking, and fingerstick is 140).
- Encourage confirmatory fingersticks before changing therapy when values seem off.
- Explain that in those moments, the algorithm is not “evil”; it is just blind.
7.2 Insulin delivery failures
No algorithm can compensate for insulin not reaching the body.
If you see:
- Rising glucose despite high or maximum autobasal, repeated auto-corrections, and no obvious carb intake → Think site failure / occlusion.
The right move is not to crank up sensitivity and ratios; it is to change the set, check for ketones, and manage like any pump failure.
8. Ethical Responsibilities: Informed Use, Not Magical Thinking
Closed-loop systems blur a line: they look like they are “in charge,” but legally and ethically, you and the patient still are.
A few hard truths:
- You cannot ethically prescribe these systems without at least a working understanding of the algorithm’s logic and limitations.
- You should not promise that “the algorithm will figure it out” for patients with chaotic eating, inconsistent use, or severe psychological barriers to self-care.
- You must address equity: these are expensive, access is uneven, and digital literacy varies wildly.
Think of the ethical duties in four buckets:
Accuracy in expectations
Tell the patient: This system will reduce your lows and smooth many highs if you do the basics (wear it, calibrate as required, bolus for meals reasonably on time). It will not remove all spikes or fully replace your decisions.Respect for autonomy but honest critique
If someone keeps overriding auto mode, running manual 90% of the time, you should ask why. Side with their lived experience, but do not collude with misunderstandings (“the pump is bad”). Review data together and interpret what the algorithm tried to do.Justice and access
Recognize that not all patients can or want to manage app updates, Bluetooth pairing, or complicated UIs. It is unethical to push the shiniest system on a patient who can barely afford strips or who lives where reliable CGM supply is a question mark.Data stewardship
These systems generate dense data. You are not expected to comb every trace manually, but you should know how to view:- Time in range
- Time below range
- Percent in auto mode
- Frequency of suspends and auto-corrections
These are not “extra.” They are now part of the basic follow-up for a patient using closed-loop insulin.
9. Translating Algorithm Logic Into What You Do in Clinic
Let me make this brutally practical. You have 15–20 minutes. Here is the mental workflow I have seen work consistently.

| Step | Description |
|---|---|
| Step 1 | Open device report |
| Step 2 | Check auto mode percent |
| Step 3 | Ask why auto is off |
| Step 4 | Review time in range and lows |
| Step 5 | Address trust, alarms, usability |
| Step 6 | Look at overnight control |
| Step 7 | Consider weakening basal or ISF at night |
| Step 8 | Examine post meal patterns |
| Step 9 | Strengthen carb ratio / timing |
| Step 10 | Fine tune targets or activity settings |
| Step 11 | Auto mode >80 percent? |
| Step 12 | Frequent suspends? |
| Step 13 | High after meals? |
Key habits:
Always look at percent of time in auto / closed loop. High-tech system that is in manual mode half the time = not really a closed loop clinically.
Separate analysis into:
- Overnight / fasting pattern
- Postprandial pattern
- Activity-related swings
When something looks wrong, ask:
Is this a parameter issue (ratios, action time)?
A behavior issue (late boluses, taking pump off)?
A sensor/infusion set issue?
Then make one or two targeted changes. Not ten. Algorithms hate chaos.
10. What You Absolutely Should Be Able to Explain to a Patient
If you can do these without looking anything up, you are ahead of most generalists and frankly some specialists.
You should be able to answer, in plain language:
“How does this system decide how much insulin to give me every 5 minutes?”
Answer: It looks at your CGM, your trend, how much insulin it thinks is still working, and your target. It then slightly increases or decreases insulin every few minutes to nudge you toward that target safely.“Why did it suspend me last night and then I woke up high?”
Answer: It predicted your glucose would drop too low and shut off insulin to protect you. The rebound high was the tradeoff for avoiding a dangerous low. We can sometimes adjust settings to reduce that see-saw.“If it is so smart, why do I still have to bolus for food?”
Answer: Because our current insulin is slow. By the time the system sees your glucose rising from food, it is already late. Your pre-meal bolus starts insulin working earlier so the algorithm has a chance to fine-tune rather than chase.“Can I just set the target to 90 mg/dL to get a 5.5 percent A1c?”
Answer: With current insulin and safety rules, that usually leads to more lows and more alarms. The systems are designed to balance safety and control. We can push tighter in some scenarios, but we need to look at your data and hypoglycemia risk carefully.
If you cannot give those answers yet, that is your personal development task. Not optional for modern diabetes care.
11. Where This Is Going – And Why You Need To Keep Up
Closed-loop insulin is not a passing fad. We are heading toward:
- More adaptive, personalized algorithms that quietly update your parameters.
- Dual-hormone systems (insulin + glucagon or insulin + amylin analogues).
- Faster insulins and alternative delivery routes that will make full closed-loop, including meals, more realistic.
- Algorithm updates pushed like phone OS updates, changing behavior overnight.
This raises new ethical and practical questions:
- What consent is meaningful when an overnight software update subtly changes control behavior?
- Who is liable when a bug slips through—a clinician, a manufacturer, neither?
- How do we explain “version 3.2” of a control algorithm change to a patient who just wants steady sugars?
You do not have to be an engineer. But you cannot afford to be algorithm-illiterate. Not anymore.
With a solid grasp of how these systems think, when they are right, when they are wrong, and what knobs you actually control, you are no longer a spectator to the technology. You are part of it. And that sets you up for the next wave of bio-digital tools that will not even bother to wait for you to catch your breath.
But that is a story for another day.
FAQ
1. Do I need to know the exact algorithm (PID vs MPC) for each device I prescribe?
No, you do not need the math, but you do need the behavior. At minimum, know: whether it predicts future glucose, whether it auto-corrects above basal, what its targets and safety limits are, and which settings the clinician can adjust. That lets you explain patterns and make rational changes without pretending it is a black box.
2. Are closed-loop systems appropriate for all patients with type 1 diabetes?
No. Patients who are unable or unwilling to wear a CGM consistently, who have severe tech aversion, or who lack basic support for troubleshooting may not benefit, and might even be harmed by frequent system failures and frustration. Closed loop is powerful for motivated users with at least minimal comfort with devices, or with strong family support. It is not a moral failing if someone opts for simpler therapy.
3. How should I adjust therapy when a patient transitions from MDI or standard pump to closed-loop?
Expect lower basal requirements and reduced correction bolus needs over time as nocturnal and between-meal control smooths out. Do not aggressively tighten A1c targets in the first weeks; focus on hypoglycemia reduction, time in range, and learning how the system behaves. Review data at 2–4 weeks, then adjust carb ratios, insulin action time, and activity/sleep modes in small steps.
4. What red flags in device data should prompt urgent intervention?
Watch for sustained time >300 mg/dL with ketones, especially if autobasal is maxed out—think infusion set failure. Frequent or prolonged pump suspensions with large morning rebounds may require immediate parameter review and education. Recurrent unrecognized hypoglycemia, despite the system’s safety logic, suggests mis-set sensitivity parameters, sensor inaccuracies, or patient behaviors (e.g., stacking manual boluses) that must be addressed quickly.


















